Illustrations by the author, Dr. Thomas T. Yamashita (except where noted otherwise)
Farmers and advisors frequently make the statement that healthy plants appear to tolerate or resist disease better than unhealthy plants. A skeptic might counter by saying that the plant is unhealthy because of disease, or the plant is healthy because it is free of disease. The two statements are similar in the obvious sense but very much different in the underlying message. The skeptic assumes a simple cause and effect relationship and focuses on the pathogen as the primary object of concern. On the other hand, observant farmers and advisors instill hope, and suggest taking proactive steps which can provide control over the health of the plant.
As it turns out, in the great majority of diseases, the fate of the plant is not solely determined by the presence of the classic disease triangle: pathogen, environmental conditions, and susceptible host. Rather, much emphasis must be placed on qualifying and characterizing the nebulous metes and bounds of a susceptible host. Many farmers and advisors say that a healthy host is less likely to be infected by the pathogen. Thus, when infection does occur, it is logical to compare key characteristics between infected and uninfected hosts.
I have long emphasized the fact that over 90% to 95 % of all examined plant diseases can be traced back to suboptimal nutrition. More specifically, one of the common factors in the chain of chronological steps leading to malady or infection resides in imbalanced nutrition. Superior nutrition helps to impart tolerance and resistance to various diseases. In this article, we will examine in detail the concepts of induced resistance, and some of the physical and physiological events that occur when a pathogen initially attacks a plant, and the make-or-break events that determine whether infection occurs.
Disease resistance in a plant is a physiological, rate-related response.
A plant’s resistance response to pathogen intrusion depends upon how quickly the plant can mount a defense. The speed of the response determines whether a plant remains healthy or becomes afflicted with disease (see Figure 1).
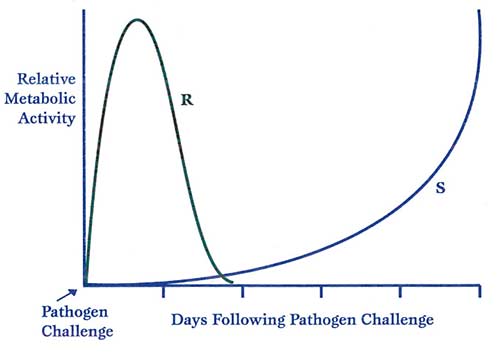
Figure 1. The rise in defense metabolism following pathogen challenge. S = a susceptible plant, R = a resistant plant. As we continue this discussion, it will become clear that rapidity of the resistance response is commensurate with the health status of the plant and its ability to quickly prioritize production of various ‘secondary metabolites’ and compounds necessary for resisting disease. All such reactions require a plant to be metabolizing nutrients efficiently, an ample supply of the molecules necessary for building tissue structures, and a supply of chemically stored energy to drive these reactions.
Plants utilize various mechanisms to literally wall off pathogens and physically prevent entrance or progress of the pathogenic intruder.
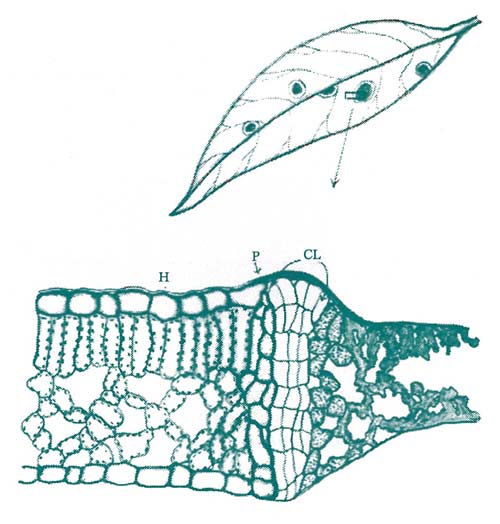
Figure 2. Infections by various agents, including fungi, bacteria, and some viruses and nematodes, will oftentimes elicit the formation of a cork layer. This cork layer is high in suberin, a fatty substance with water repellent and tissue-protective qualities. The cork layer acts to prevent the flow of nutrients to the infection site, and seals the wound following sloughing of the invaded tissue. (Key: H = healthy area, P = phellogen, CL = cork layer, I = infected area.) Redrawn from Cunninghan, 1928.
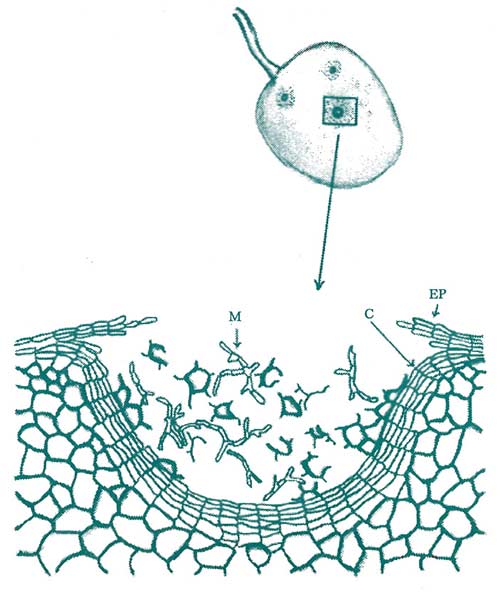
Figure 3. Fruit tissues will also respond with the formation of a cork layer. The drawing above depicts the walling off of a fungal pathogen with rapid and timely cork layer formation. (Key: M = mycelium of the fungal pathogen, C = cork layer, Ep = epidermis.) Redrawn from G.E. Ramsey, 1917.
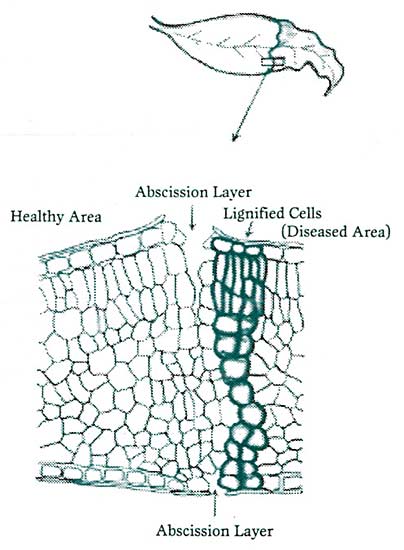
Figure 4. Many plant tissues invaded by a pathogenic agent will quickly wall off the organism’s further advancement by first forming an impregnable layer of lignified cells. This effectively cuts further movement of nutrients to the infected side and the pathogen. This is quickly followed by an adjacent abscission layer, which allows the diseased portion to slough off, distancing the pathogen’s proximity from healthy areas of the tissue. Redrawn from Samuel, 1927.
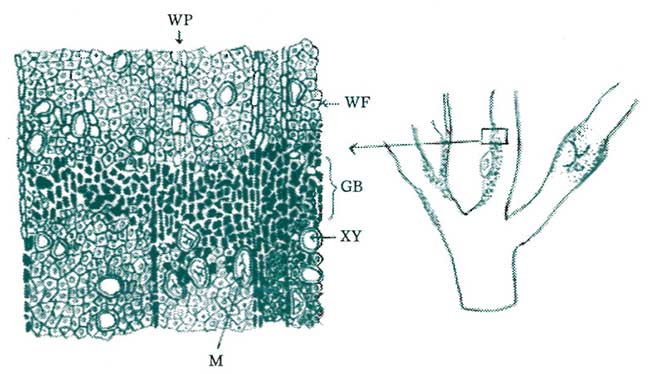
Figure 5. When woody tissues are invaded, isolation or wailing off the intruding pathogen oftentimes takes the form of gum production and lignification. Gums are complex polysaccharides with colloidal properties which allow them to expand and contract as well as to form impenetrable barriers against the advancing pathogen. Intercellular spaces surrounding the zone of infection are quickly filled with gums, thereby forming an impenetrable barrier. Adjacent tissues eventually become lignified, thereby completing the mature walled-off barrier. (Key: WP = wood parenchyma tissue, WF = wood fiber, GB = gum barrier, XV = xylem vessels, M = mycelia of the fungal pathogen.) Redrawn from Hesler, 1916.
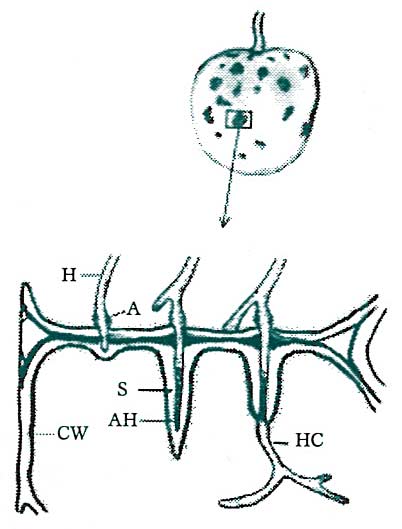
Figure 6. Attempted invasion of host tissues by a potential pathogen is often responded to with the production of ‘adjusted’ tissues. At the localized regions of invasion, cell walls thicken, swelling to many times normal density. Furthermore, invaded portions of the pathogen are ensheathed by extensions of the host cell wall. (Key: H = pathogen hyphae, A = pathogen appresorium or “pad,” HC = hypha in the host cytoplasm, AH = advancing hypha, S = sheath formed by the thickened cell wall of the host, CW = cell wall of the host.) Redrawn from Agrios, 1978.
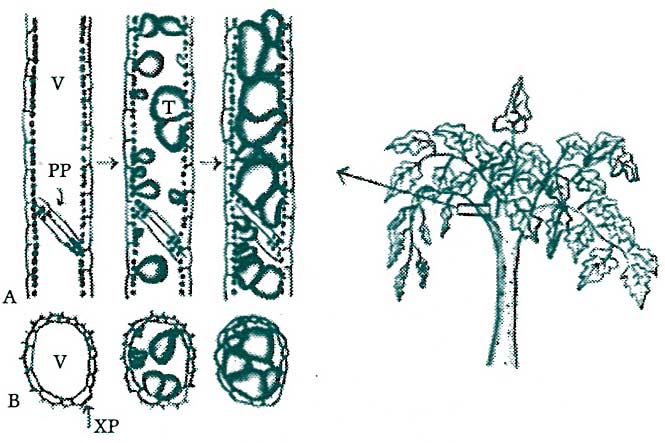
Figure 7. Many vascular pathogens are localized within the initially invaded region of the host by the formation of ‘tyloses.’ Tyloses are cellulosic outgrowths from adjacent parenchyma cells, which grow through the perforated conductive vessels, interrupting the active and passive transport of the pathogen from the site of local invasion to outlying areas of host tissue. (Key: V = xylem vessel, PP = perforated vessel plate, T = tylose, XP = xylem parenchyma Cell.)
(A) depicts longitudinal sections of a healthy, noninvaded xylem vessel (far left), the beginning of tylose formation following invasion (middle), and advanced tylose formation (far right).
(B) depicts conditions illustrated in (A) but in transverse sections. Redrawn from Agrios, 1978 by TTY.
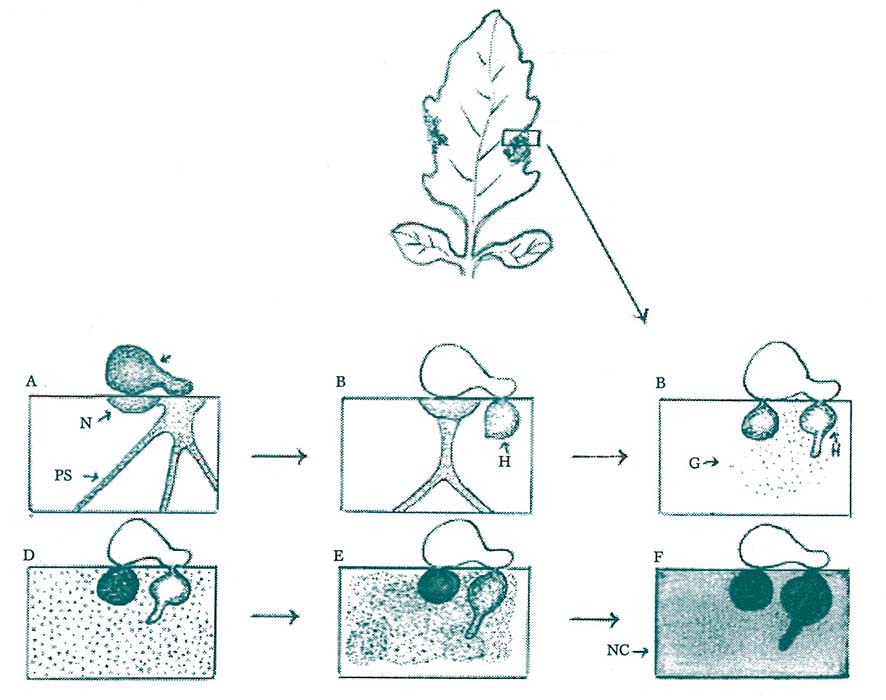
Figure 8. In many plant hosts invaded by an obligate parasite (e.g. late blight or plant-parasitic nematodes), the localized tissue walls off the intruder by quickly self-destructing, limiting further sustenance of the pathogen, which depends on the presence of living tissue. This is often referred to as a hypersensitive response. The typical steps in this reaction include: a) Movement of the host cell nucleus towards the invading pathogen, b) Disintegration of the nucleus, c) Formation of dark, resin-like granules in the cytoplasm, d) Death of the cell and successive degeneration of the invading pathogen. (Key: S = pathogen spore, N = host cell nucleus, PS = protoplasmic strand, H = pathogen hypha, G = cytoplasmic granules, NC = necrotic cell of the host. Redrawn from K. Tomiyama, 1956.
The above illustrations of various physical defense responses are intended to provide us with a sound mental picture of the walling-off phenomenon.
They also provide us with a model for understanding the underlying reasons as to why a healthy plant hosts more natural resistance to various pathogens than does an unhealthy plant. There are multiple forms of resistance to pathogens, and the walling-off phenomenon is one of the more common mechanisms.
In many cases the host plant either has a specific biochemical make-up which is not conducive to growth or even germination of a pathogenic agent. In other instances, the host may lack biochemical components which are necessary for infection and growth of the pathogen. What we are covering in the walling-off phenomenon examples represents reactions of potentially infective host-pathogen interactions (PIHPI), and the characterization and intricacies of these interactions as they relate to a successful resistance response.
In the greatest majority of PIHPI, there is the need for a sudden and timely rise in host metabolism to mount a defense reaction (see Fig. 1). This sudden increase in host metabolism is generally directed towards the production of agents of resistance, some of which are physical, walling-off compounds. Many of the agents of resistance are biochemical antagonists, which for various reasons create a suboptimal or harsh environment for growth and reproduction of the invader. The latter are often referred to as ‘secondary metabolites,’ as they are most commonly formed in response to invasion, and not necessarily ahead of invasion. They are also called secondary metabolites because these compounds are not normally found in an unchallenged host at high concentrations, but instead are temporarily formed to ward off a potential pathogen. Thus, both the walling-off compounds and secondary metabolites are reactionary compounds, formed as you might say a counter-punch to potential invasion of the host.
The host biochemistry which produces the various compounds of resistance must be efficient. Furthermore, the production of compounds of resistance necessarily involves both biochemical conversions of existing carbon molecules, as well as de novo synthesis (original production) of parent carbon molecules. Efficient machinery is necessary to produce the various enzyme-mediated reactions needed at a velocity sufficient for an effective immune response. The higher the velocity of these reactions, the more compounds of resistance are produced on a timely basis and the more effectively potential pathogens are warded off.
Understanding the resistance response of plants requires a closer examination of walling-off compounds and secondary metabolites.
It’s often easy to react with shock when informed of the cost of a piece of custom-built furniture. But when informed of the cost of materials and the intricate, detailed steps necessary to build that furniture, the price tag suddenly looks more reasonable. This is the same approach we need to take with the resistance response. Thus, let’s take a brief look at just a few of the components that must be ‘built’ to mount a successful response.
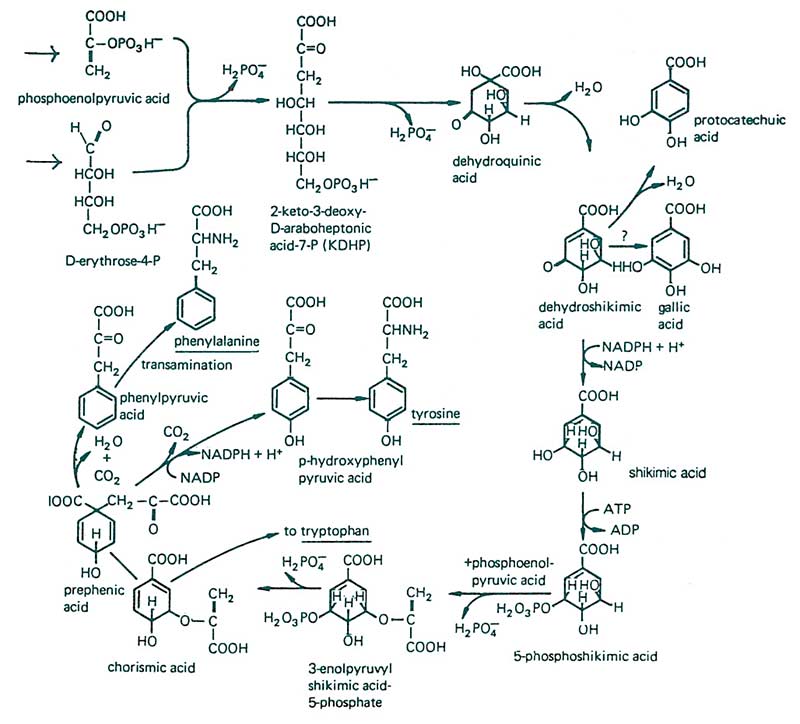
Figure 9. A good starting point for compound of resistance production begins with understanding the specifics of secondary metabolite formation. Phosphoenolpyruvic acid comes from the glycolytic pathway, and erythrose 4-phosphate from either the pentose phosphate pathway or the ‘Calvin cycle.’ Various reactions take place producing the parent molecules (tryptophan, tyrosine and phenylalanine), which are precursors for the production of various secondary metabolites. This set of reactions is also known as the ‘shikimic acid pathway.’ From F.B. Salisbury and C.W. Ross, 1985.
From the precursor molecules (Tryptophan, Tyrosine & Phenylalanine) arise a number of secondary metabolites instrumental in the resistance response:
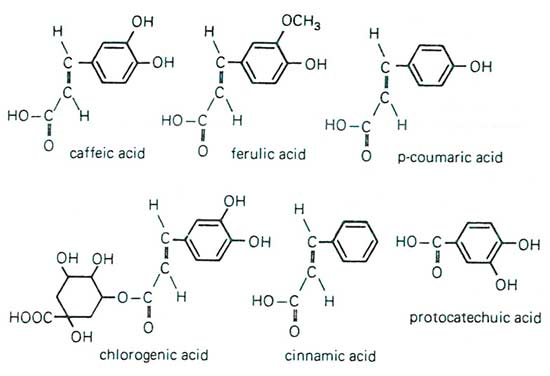
Figure 10. Miscellaneous simple phenols and related compounds such as caffeic acid, ferulic acid, coumaric acid, chlorogenic acid, cinnamic acid, and protocatechuic acid are not only known for their direct part in resistance to pathogens, but are also precursors to further derivatives instrumental in imparting the resistance response. From F.B. Salisbury and C.W. Ross, 1985.
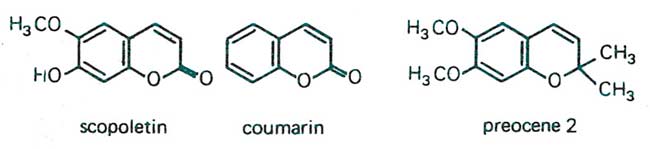
Figure 11. Close relatives of the simple phenolic acids are the coumarins, which are formed via the shikimic acid pathway from phenylalanine and cinnamic acid. Shown here are scopoletin, coumarin, and preocene. Scopoletin, for example, is found in the seed coat of various plant species, imparting protection from invaders as well as causing seed dormancy. Preocene occurs in the floss flower (Ageratum houstonianum) and is known to cause premature metamorphosis in various insect pest species by decreasing the level of insect juvenile hormone, inducing formation of sterile adults. Coumarin is the volatile compound formed when alfalfa is cut for harvest and is responsible for the fragrance of cut hay. It is, among others, instrumental in protecting the wounded tissue until rapid cork tissue and lignification occur at the cut ends. From W.S. Bowers et. al., 1976.
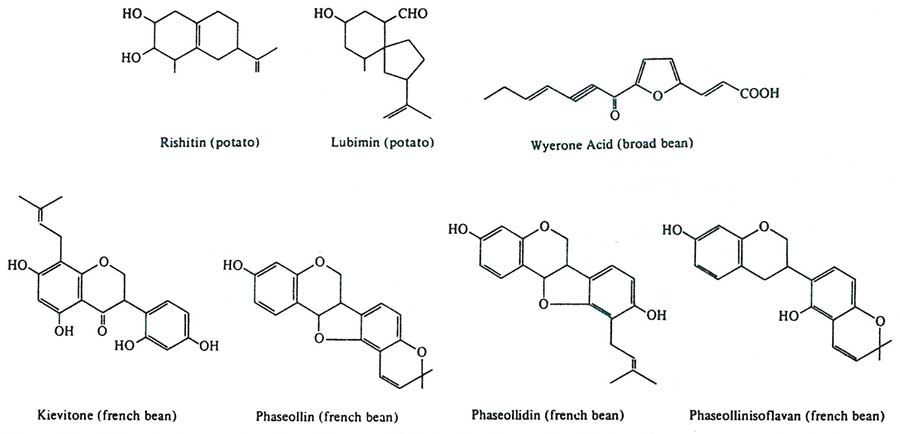
Figure 12. Phytoalexins are phenolic and isoprenoid derivatives known for their broad-spectrum protection in the resistance response. The word is taken from the Greek, “phyton” for ‘plant,’ and “alexin” for ‘warding-off substance.’ Shown above are some phytoalexins observed in the potato (rishitin and lubimin), broad bean (wyerone acid), French bean (kievitone, phaseollin, phaseollidin and phaseollinisoflavan). For example, rishitin and lubimin are instrumental in potato varieties resistant to Phytophthora infestans (potato late blight fungus). Kievitone and phaseollin impart resistance in French beans to Rhizoctonia solani. From J. Friend, 1981.
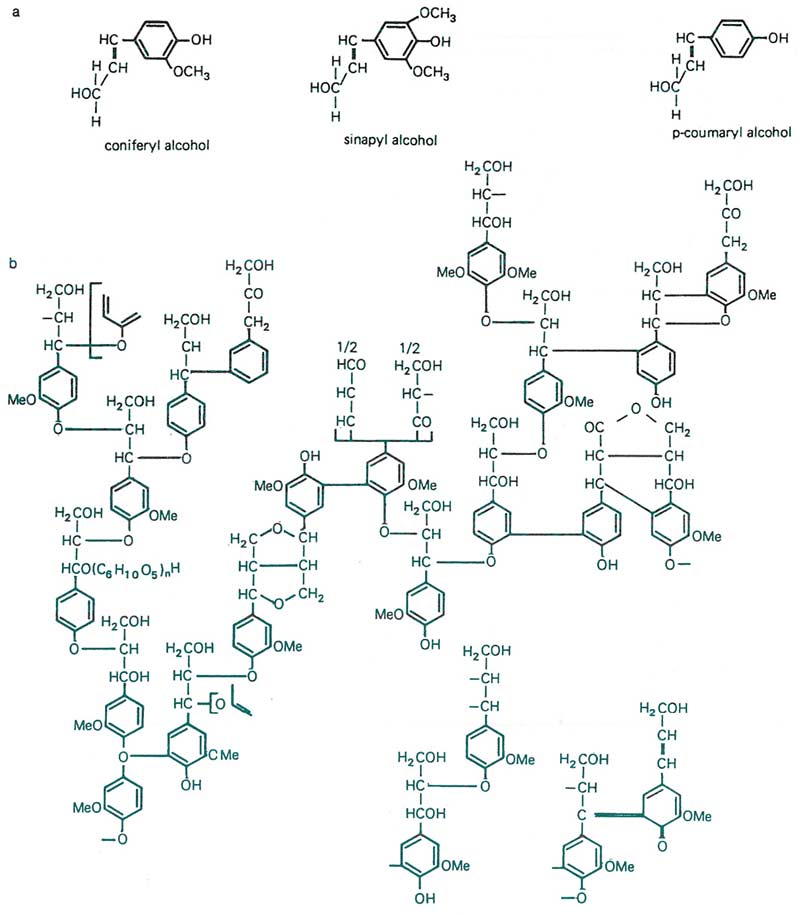
Figure 13. Next to cellulose, lignin is one of the most abundant natural compounds in the plant world. There are few organisms with the ability to digest lignin, one of which are the white rot mushroom fungi. Thus, impregnation of tissues with lignin promotes resistance to a broad spectrum of organisms. It also imparts tissue stability and cold tolerance. (a) Depicts common subunits found in lignins, and (b) illustrates the complexity of the lignin molecule, shown here by spruce lignin, with an alcohol ratio of 14 coumaryl : 80 coniferyl : 6 sinapyl. From K. Freudenberg, 1965.
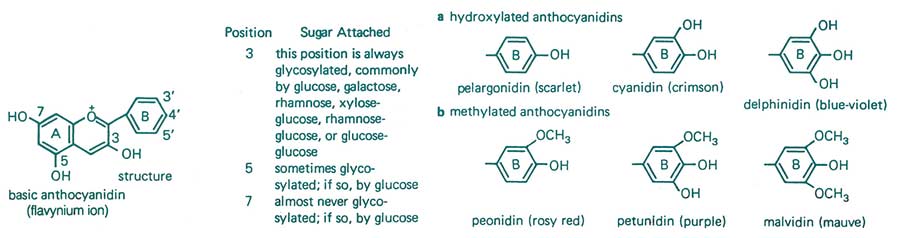
Figure 14. Flavanoids round out our discussion on secondary metabolites associated with imparting natural resistance to disease. Flavanoids are 15-carbon ringed compounds with varying levels of double bonds, endowing them with the ability to absorb varying levels of visible light. They are responsible for the unique pigmentation of plants. The basic flavynium ion can be substituted by various (a) hydroxyl or (b) methyl groups. Flavanoids are believed to be involved in resistance to diseases and insects. However, some of these chemicals, such as anthocyanins, also play a major role in helping to maintain plant health through protection against the weakening effects of ultraviolet light, such as the inactivation of chlorophyll. From F.B. Salisbury and C.W. Ross, 1985.